Chiral Bidentate Phosphoramidite-Pd Catalyzed Asymmetric Decarboxylative Dipolar Cycloaddition for Multistereogenic Tetrahydrofurans with Cyclic N‑Sulfonyl Ketimine Moieties
Hao-Peng Lv1, Xiao-Peng Yang1, Bai-Lin Wang1, Hao-Di Yang1, Xing-Wang Wang1,*(王兴旺), and Zheng Wang2,*(王正)
1Key Laboratory of Organic Synthesis of Jiangsu Province, College of Chemistry, Chemical Engineering and Materials Science & Collaborative Innovation Center of Suzhou Nano Science and Technology, Soochow University, Suzhou 215123, China
2State Key Laboratory of Organometallic Chemistry, Center for Excellence in Molecular Synthesis, Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, Shanghai 200032, China
Org. Lett. 2021, 23(12), 4715--4720
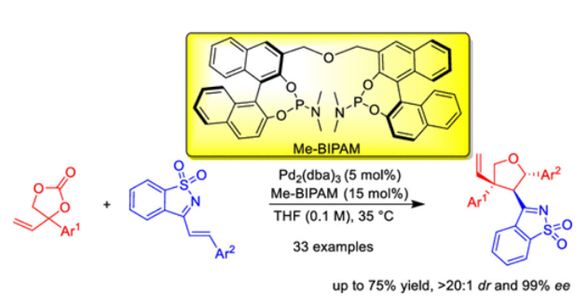
An asymmetric [3 + 2] cycloaddition of vinyl ethylenecarbonates (VECs) and (E)-3-arylvinyl substituted benzo[d] isothiazole 1,1-dioxides has been developed using the Pd complex of a bidentate phosphoramidite (Me-BIPAM) as the catalyst, providing a wide variety of chiral multistereogenic vinyltetrahydrofurans in good yields with excellent diastereo- and enantioselectivities (up to >20:1 dr, 99% ee).
链接:https://pubs.acs.org/doi/abs/10.1021/acs.orglett.1c01411
