Calcium-mediated C(sp3 )−H Activation and Alkylation of Alkylpyridines
Xizhou Zheng1, Iker del Rosal2, Xian Xu1, Yingming Yao1,*(姚英明), Laurent Maron2,*, and Xin Xu1,*(徐信)
1Key Laboratory of Organic Synthesis of Jiangsu Province, College of Chemistry, Chemical Engineering and Materials Science, Soochow University, Suzhou 215123, P. R China
2LPCNO, CNRS, and INSA, Université Paul Sabatier, 31077 Toulouse, France
Inorg. Chem.2021, 60, 5114--5121
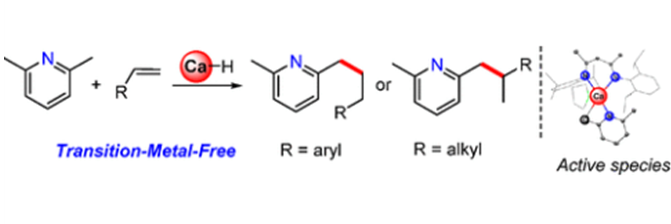
Main-group metal calcium-mediated alkylpyridine benzylic C(sp3)–H activation and functionalization have been achieved. The reaction of a calcium hydride complex [{(DIPPnacnac)CaH(thf)}2] (DIPPnacnac = CH{(CMe)(2,6-iPr2–C6H3N)}2) with two equivalents of 2,6-lutidine rapidly yields a monomeric calcium alkyl complex with the release of dihydrogen. A hydride/carbon-bridged binuclear calcium complex [{(DIPPnacnac)Ca}2(μ-H){2-Me-6-(μ-CH2)-Py}(thf)] is obtained from an equimolar treatment of calcium hydride and 2,6-lutidine that is readily converted into mono- or binuclear calcium alkyl complexes upon subsequent addition of 2,6-lutidine. DFT calculations and kinetic studies are conducted to determine their reaction profiles. More significantly, this calcium hydride complex catalyzes regioselective benzylic C–H bond addition of alkylpyridines to a variety of alkenes, affording linear or branched alkylated pyridine derivatives.
链接:https://pubs.acs.org/doi/10.1021/acs.inorgchem.1c00161
