Boosted Inner Surface Charge Transfer in Perovskite Nanodots@Mesoporous Titania Frameworks for Efficient and Selective Photocatalytic CO2 Reduction to Methane
Qi-MengSun1, Jing-JingXu2, Fei-Fei Tao2, Wen Ye3, Chang Zhou1, Jing-HuiHe1,*(贺竞辉), andJian-MeiLu1,*(路建美)
1 College of Chemistry, Chemical Engineering and Materials Science, Collaborative Innovation Center of Suzhou Nano Science and Technology, National United Engineering Laboratory of Functional- ized Environmental Adsorption Materials, Soochow University, Suzhou 215123 (P. R. China)
2 Department of Chemistry and Chemical Engineering, Shaoxing University, Zhejiang 312000 (P. R. China)
3 State Key Laboratory of Radiation Medicine and Protection, Collaborative Innovation Center of Suzhou Nano Science and Technology, National United Engineering Laboratory of Functional- ized Environmental Adsorption Materials, Soochow University, Suzhou 215123 (P. R. China)
Angew. Chem. Int. Ed. 2022, 61, e202200872 (1 of 9)
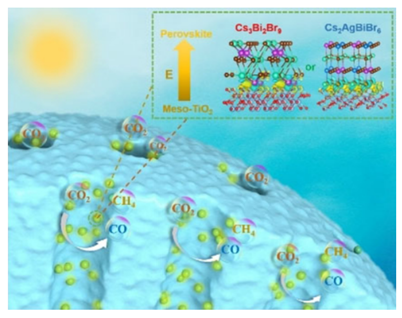
Exploring high-efficiency and stable halide perovskite-based photocatalysts for the selective reduction of CO2 to methane is a challenge because of the intrinsic photo- and chemical instability of halide perovskites. In this study, halide perovskites (Cs3Bi2Br9 and Cs2AgBiBr6) were grown in situ in mesoporous TiO2 frameworks for an efficient CO2 reduction. Benchmarked CH4 production rates of 32.9 and 24.2μmolg−1h−1 with selectivities of 88.7% and 84.2%, were achieved, respectively, which are better than most reported halide perovskite photocatalysts. Focused ion-beam sliced-imaging techniques were used to directly image the hyperdispersed perovskite nanodots confined in mesopores with tunable sizes ranging from 3.8 to 9.9nm. In situ X-ray photoelectronic spectroscopy and Kelvin probe force microscopy showed that the built-in electric field between the perovskite nanodots and mesoporous titania channels efficiently promoted photo-induced charge transfer. Density functional theory calculations indicate that the high methane selectivity was attributed to the Bi-adsorption-mediated hydrogenation of *CO to *HCO that dominates CO desorption.
链接:https://onlinelibrary.wiley.com/doi/full/10.1002/anie.202200872
