Palladium-Catalyzed Regioselective Diarylation/Deamination of Homoallylamines: Modular Assembly of 1,1,4,4-Tetraaryl-1,3- butadienes
Nan-Quan Jiang1, Tian-Shu Wu1, Min Pan1, Zhong-Jian Cai*(蔡忠建), and Shun-Jun Ji1,2,*(纪顺俊)
1 Key Laboratory of Organic Synthesis of Jiangsu Province, College of Chemistry, Chemical Engineering and Materials Science & Collaborative Innovation Center of Suzhou Nano Science and Technology, Soochow University, Suzhou 215123, China
2 Suzhou Baolidi Functional Materials Research Institute, Suzhou 215144, China
Org. Lett.2022, 24, 7465--7469
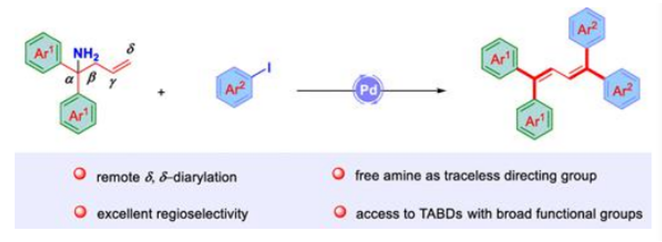
Herein, a novel palladium-catalyzed regioselective diarylation/deamination of homoallylamines is described. During the process, a ubiquitous free amine (NH2) was used as the directing group to accomplish the regioselective δ,δ-diarylation and it was removed in situ to form the privileged 1,1,4,4-tetraaryl-1,3-butadiene motif. This chelate-controlled Heck-type approach eliminates the traditional need of preinstalled and hard removable directing groups, proceeds under simple conditions, and exhibits good tolerance to a wide range of synthetically useful functional groups.
链接:https://pubs.acs.org/doi/full/10.1021/acs.orglett.2c03057
