Salt-Stabilized Silylzinc Pivalates for Nickel-Catalyzed Carbosilylation of Alkenes
Jixin Wang , Zhili Duan , Xingchen Liu , Shoucheng Dong , Kaixin Chen , Jie Li*(李杰)
Soochow Univ, Coll Chem Chem Engn & Mat Sci, Key Lab Organ Synth Jiangsu Prov, Ren Ai Rd 199, Suzhou 215123, Peoples R China
Angew. Chem.Int. Ed. 2022,61, e202202379
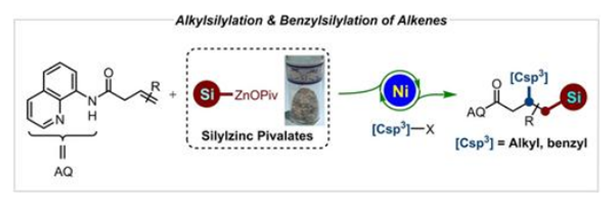
We herein report the preparation of solid and salt-stabilized silylzinc pivalates from the corresponding silyllithium reagents via transmetalation with Zn(OPiv)(2). These resulting organosilylzinc pivalates show enhanced air and moisture stability and unique reactivity in the silylative difunctionalization of alkenes. Thus, a practical chelation-assisted nickel-catalyzed regioselective alkyl and benzylsilylation of alkenes has been developed, which provides an easy method to access alkyl silanes with broad substrate scope and wide functional group compatibility. Kinetic experiments highlight that the OPiv-coordination is crucial to improve the reactivity of silylzinc pivalates. Furthermore, late-stage functionalizations of druglike molecules and versatile modifications of the products illustrate the synthetical utility of this protocol.
链接:https://onlinelibrary.wiley.com/doi/full/10.1002/anie.202202379
