Directly anchoring Fe3C nanoclusters and FeNx sites in ordered mesoporous nitrogen-doped graphitic carbons to boost electrocatalytic oxygen reduction
Zhi Chen a, Xingmin Gao a, Xiangru Wei a, Xinxia Wang b, Yanguang Li b, TaoWu c,Jun Guo d, Qinfen Gu e, Winston Duo Wu a, Xiao Dong Chen a, Zhangxiong Wu a, *(吴张雄),Dongyuan Zhao f, g
a Suzhou Key Laboratory of Green Chemical Engineering, School of Chemical and Environmental Engineering, College of Chemistry, Chemical Engineering and Materials Science, Soochow University, Suzhou, Jiangsu 215123, China
b Institute of Functional Nano and Soft Materials (FUNSOM), Soochow University, Suzhou, Jiangsu 215123, China
c School of Chemistry, College of Chemistry, Chemical Engineering and Materials Science, Soochow University, Suzhou, Jiangsu 215123, China
d Testing and Analysis Centre, Soochow University, Suzhou, Jiangsu 215123, China
e Australian Synchrotron, 800 Blackburn Road, Clayton, Victoria 3168, Australia
f Department of Chemistry and Laboratory of Advanced Materials, Fudan University, Shanghai 200433, China
g Department of Chemical Engineering, Monash University, Clayton, Victoria 3800, Australia
Carbon 121 (2017) 143--153
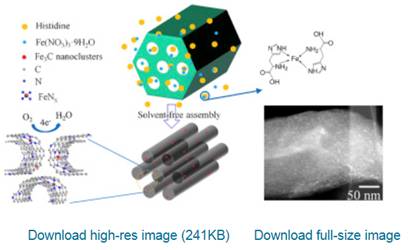
Porous carbon materials doped with nano-sized transition metal carbides and/or metal-nitrogen coordinative sites are promising oxygen reduction electrocatalysts. The doping of such functionalities in carbon materials with desirable concentration, ultra-small size and stable configuration is still a challenge. In this paper, by grinding and pyrolyzing solid mixtures of an amino acid, an iron salt, and a mesoporous silica template, we demonstrate a solvent-free assembly approach to directly anchor both Fe3C nanoclucters and FeNx sites into nitrogen-doped ordered mesoporous graphitic carbon materials. The carbonaceous electrocatalysts are imparted with several fascinating features, namely, highly dispersed ultra-small Fe3C nanoclusters of 1-3 nm, well-anchored FeNx sites, nitrogen-doped well-graphitized carbon frameworks, and ordered mesopores (similar to 5.4 nm) and high surface areas (>1000 m(2)/g), respectively. The combination of these features makes these electrocatalysts exceptional for oxygen reduction reaction under both alkaline and acidic electrolytes, i.e. superior catalytic activities (e.g. onset and half-wave potentials up to 1.00 and 0.89 V vs. the reversible hydrogen electrode in alkaline solution), outstanding stabilities and excellent methanol tolerance, respectively. An in-depth study has been conducted to identify and characterize the key active sites in these electrocatalysts and to elucidate several important influencing factors to optimize the catalytic performance. (C) 2017 Elsevier Ltd. All rights reserved.
链接:http://www.sciencedirect.com/science/article/pii/S0008622317305365
