Copper-catalyzed construction of eightmembered rings via oxidative ring expansion and intermolecular cyclization sequencing of indoles with amidines: efficient synthesis of benzo[1,3,5]triazocin-6(5H)-ones†
Wen-Bin Cao a, Xue-Qiang Chu a, Yu Zhou a, Ling Yin ab, Xiao-Ping Xu*a (徐小平)and Shun-Jun Ji *a(纪顺俊)
a Key Laboratory of Organic Synthesis of Jiangsu Province, College of Chemistry, Chemical Engineering and Materials Science & Collaborative Innovation Center of Suzhou Nano Science and Technology, Soochow University, Suzhou,215123, P. R. China.
b Department of Chemistry and Chemical Engineering, Jining University,
Qufu 273155, P. R. China
Chem. Commun.,2017, 53, 6601--6604
Novel and efficient synthesis of eight-membered nitrogen heterocycles in one pot by copper-catalyzed oxidative ring expansion and intermolecular cyclization sequencing of indoles with amidines has been described. This protocol features easily available substrates and high step-economy. More importantly, dioxygen as the most ideal oxidant was employed under mild reaction conditions. A variety of valuable benzo[1,3,5]triazocin-6(5H)-one derivatives bearing functional groups were assembled in middle to good yields.
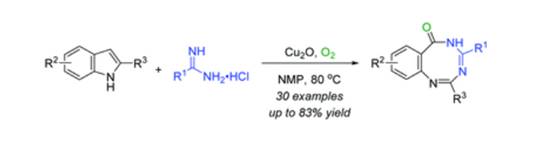
链接:http://pubs.rsc.org/en/Content/ArticleLanding/2017/CC/C7CC02815F#!divAbstract
