Aromatic diselenide crosslinkers to enhance the reprocessability and self-healing of polyurethane thermosets†
Xiaowei An‡a, Robert H. Aguirresarobe‡b, Lourdes Irusta b, Fernando Ruipérez b,Jon M. Matxain c, Xiangqiang Pan a, Nora Aramburu,b David Mecerreyes b,d,Haritz Sardon *b,d and Jian Zhu *a(朱健)
a Jiangsu Key Laboratory of Advanced Functional Polymer Design and Application,College of Chemistry, Chemical Engineering and Materials Science, Soochow University, Suzhou, 215123, PR China.
b POLYMAT, University of the Basque Country UPV/EHU, Joxe Mari Korta Center,Avda. Tolosa 72, 20018 Donostia-San Sebastián, Spain.
c Kimika Fakultatea, Euskal Herriko Unibertsitatea UPV/EHU and Donostia International Physics Center (DIPC), P.K. 1072, 20080 Donostia, Spain
d IKERBASQUE Basque Foundation for Science, Bilbao, Spain
Polym. Chem., 2017, 8, 3641--3646
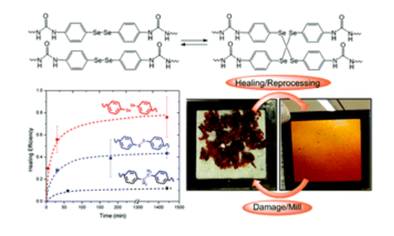
Dynamic structures containing polymers are considered a new class of polymeric materials with very attractive properties, since they can behave as thermosets at room temperature but at the same time they can be reprocessed to any shape. Recently, aromatic disulfides have been explored as dynamic bonds because of their good exchange reaction rate at low temperatures. In this work, we explore the utilization of aromatic diselenides on the basis of their lower bond energies in comparison with their disulfide counterparts. By comparing a model exchange reaction between aromatic diselenides and aromatic disulfides, the ability of aromatic diselenides to exchange faster than aromatic disulfides has been demonstrated. These results are supported by molecular quantum chemical calculations, confirming the faster exchange of aromatic diselenides in comparison with disulfides. In addition, a [2 + 1] radical-mediated reaction mechanism is proposed for aromatic diselenides to understand their faster kinetic exchange. An aromatic diselenide has been incorporated into polyurethane networks using a para-substituted amine diphenyl-diselenide. The resulting materials not only exhibit faster self-healing properties than the corresponding disulfide based materials, but also show the ability to be processed at temperatures as low as 100 °C. The fact that aromatic amines are already used as crosslinkers in a wide range of commercial products makes this system very attractive.
链接:http://pubs.rsc.org/en/content/articlelanding/2017/py/c7py00448f#!divAbstract
