Chiral N-Heterocyclic Carbene-Catalyzed Asymmetric Michael–Intramolecular Aldol-Lactonization Cascade for Enantioselective Construction of β-Propiolactone-Fused Spiro[cyclopentane-oxindoles]
Jun-Qi Zhang, Nai-Kai Li, Shao-Jie Yin, Bing-Bing Sun,Wei-Tai Fan,Xing-Wang Wang*(王兴旺)
Key Laboratory of Organic Synthesis of Jiangsu Province, College of Chemistry, Chemical Engineering and Materials Science, Soochow University, Suzhou, People's Republic of China
Advanced Synthesis & Catalysis, Volume 359, Issue 9 May 2, 2017Pages 1541--1551
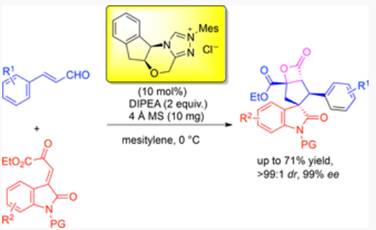
A cascade asymmetric Michael–intramolecular aldol-lactonization of enals with oxindolyl β,γ-unsaturated α-keto esters was developed. An optically pure aminoindanol-derived triazolium-based N-heterocyclic carbene was used as the catalyst. The corresponding desired β-propiolactone-fused spiro[cyclopentane-oxindoles] were obtained in moderate yields with excellent diastereoselectivities and enantioselectivities. Notably, the obtained enantio-enriched highly functionalized complex molecules contain four contiguous stereocenters, including a spiro all-carbon center and a quaternary carbon center.
链接:http://onlinelibrary.wiley.com/doi/10.1002/adsc.201601259/abstract
