Cyanotrifluoromethylthiolation of Unactivated Olefins through Intramolecular Cyano Migration
Meishan Ji1, Zhen Wu1, Jiajia Yu1, Xiaobing Wan1,*(万小兵) andChen Zhu1,2,*(朱晨)
1Key Laboratory of Organic Synthesis of Jiangsu Province, College of Chemistry, Chemical Engineering and Materials Science, Soochow University, Jiangsu, People's Republic of China
2Key Laboratory of Synthetic Chemistry of Natural Substances, Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, Shanghai, People's Republic of China
Advanced Synthesis & Catalysis,Volume 359, Issue 11, pages 1959--1962, June 6, 2017
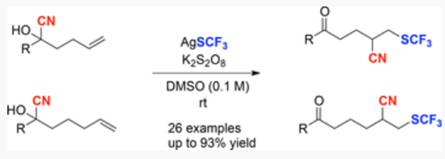
A new protocol for the elusive cyanotrifluoromethylthiolation of unactivated olefins is described. A broad range of vicinal CF3S-substituted alkyl nitriles are furnished in good yields under mild reaction conditions. The intramolecular cyano migration strategy is harnessed for the trifluoromethylthiolation of alkenes for the first time, thus offering an efficient tool for the construction of alkyl trifluoromethylthioethers.
链接:http://onlinelibrary.wiley.com/doi/10.1002/adsc.201700218/abstract
