Synthesis and Reactivity of Triangular Heterometallic Complexes Containing Zn−Zn Bond
Shengjie Jiang1, Yanping Cai1, Ambre Carpentier2, Iker del Rosal2, Laurent Maron2,*, and Xin Xu1,*(徐信)
1 Key Laboratory of Organic Synthesis of Jiangsu Province, College of Chemistry, Chemical Engineering and Materials Science, Soochow University, Suzhou 215123, P. R. China
2 LPCNO, CNRS & INSA, Université Paul Sabatier, 31077 Toulouse, France
Inorg. Chem.2022, 61, 8083--8089
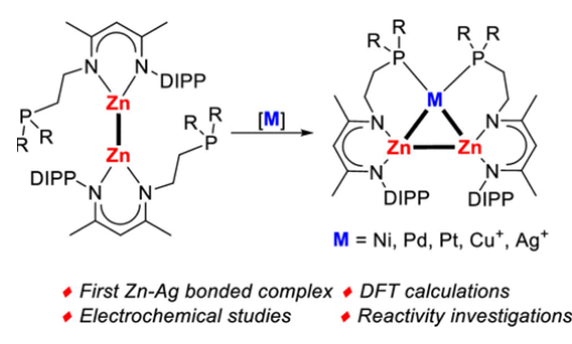
This work provides a facile access to a series of triangular [Zn2M] (M = group 10 and 11 metals) clusters. Treatment of Zn–Zn-bonded compounds [LZn–ZnL] (L = CH3C(2,6-iPr2C6H3N)CHC(CH3)(NCH2CH2PR2); R = Ph, iPr) with zero-valent transition-metal reagents selectively afforded the corresponding triangular clusters [Zn2M], where M = Ni(0), Pd(0), and Pt(0). Notably, the isoelectronic triangular clusters [Zn2M]+, where M = Ag(I) and Cu(I), could also be obtained by reactions of [LZn–ZnL] with AgOTf and CuOTf, respectively. The [Zn2Ag]+ complex containing elusive Zn–Ag bonds was investigated by density functional theory analysis, showing a 3c–2e bonding feature in the metallic ring. The electrochemical behaviors of [Zn2M] complexes were examined and revealed the donation of electron density from the Zn–Zn σ-bond to the metal centers. Reaction of the [Zn2Ni] complex with isocyanide gave heterometallic species by coordination of isocyanide to the nickel center, keeping the trimetallic ring core structure intact. In contrast, the Zn–Zn bond was rapidly cleaved upon treatment of the [Zn2Ni] complex with dihydrogen or phenyl acetylene, generating the hydride- or acetylide-bridged heterotrimetallic complex.
链接:https://pubs.acs.org/doi/10.1021/acs.inorgchem.2c0095
