Selenolactone as a Building Block toward Dynamic Diselenide-Containing Polymer Architectures with Controllable Topology
Xiangqiang Pan*†‡(潘向强), Frank Driessen‡, Xiulin Zhu†, and Filip E. Du Prez*‡
† Jiangsu Key Laboratory of Advanced Functional Polymer Design and Application, Department of Polymer Science and Engineering, College of Chemistry, Chemical Engineering and Materials Science, Soochow University, Suzhou 215123, P.R. China
‡ Department of Organic and Macromolecular Chemistry, Polymer Chemistry Research Group, Ghent University, Krijgslaan 281, S4-bis, B-9000 Ghent, Belgium
ACS Macro Lett., 2017, 6 (2), pp 89–92
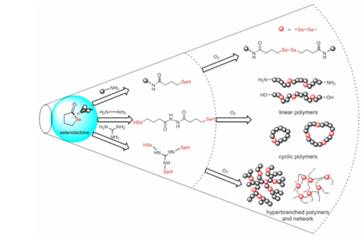
A versatile protocol for the synthesis of a variety of multiresponsive diselenide-containing polymeric architectures was investigated. It consists of a one-pot, two-step process with the generation of a selenol by in situ nucleophilic ring opening of selenolactone with a broad range of amine-containing structures, followed by the transformation of the obtained compounds to the corresponding diselenide through a spontaneous oxidation coupling reaction. After elaboration of this one-pot reaction, a number of routes based on selenolactones have been developed for the successful synthesis of functional, linear, branched, cyclic, and cross-linked polymers via a mild, straightforward process. Moreover, the polymer end groups can be easily modified by changing the ratio of amine and selenolactone or sequential Michael addition of selenol to the methacrylic ester. At last, the self-healing properties of the diselenide-containing networks were determined by exposing a cut sample of the polymer to UV light.
链接:http://pubs.acs.org/doi/abs/10.1021/acsmacrolett.6b00944
