Highly Active, Selective, and Stable Direct H2O2Generation by Monodispersive Pd–Ag Nanoalloy
Jin Zhang†,Bolong Huang*‡,Qi Shao†, andXiaoqing Huang*†(黄小青)
†College of Chemistry, Chemical Engineering and Materials Science,Soochow University,Suzhou,Jiangsu215123,China
‡Department of Applied Biology and Chemical Technology,The Hong Kong Polytechnic University, Hung Hom,Kowloon,Hong Kong SAR,China
ACS Appl. Mater. Interfaces,2018,10(25), 21291--21296
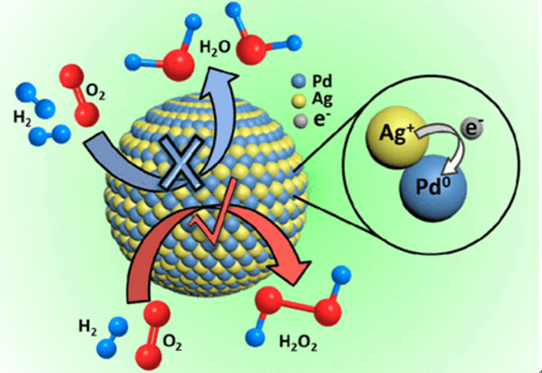
Hydrogen peroxide (H2O2), a green oxidant, has wide applications in various chemical syntheses and is also a promising candidate to replace the traditional toxic oxidants. The direct synthesis of H2O2from H2and O2is a potential approach, as it is a green and atomically economic reaction. However, the most previous systems are notorious in complicated post-purification procedures, high energy cost, and low selectivity because of the uncontrollable O–O bond cleavage. We have solved this challenge by tuning the chemical state of Pd with high H2O2productivity of 80.4 mol kgcat–1h–1and high H2O2selectivity of 82.1% via the design of Pd–Ag nanoalloys with flexibly tuned size and composition. The created Pd–Ag nanoalloy also exhibits excellent stability with limited performance decay over recycles. The X-ray photoelectron spectroscopy analysis confirms the electron transfer from Ag to Pd, which generates more Pd0and enables improved H2O2productivity. The theoretical calculation shows that the incorporation of Ag into Pd is beneficial for the stabilization of O22–and the cleavage of H2for the enhanced H2O2generation. In addition, the enhanced H2O2desorption on Pd–Ag nanoalloy is beneficial for releasing H2O2, which results in the increased H2O2selectivity.
链接:https://pubs.acs.org/doi/10.1021/acsami.8b03756
