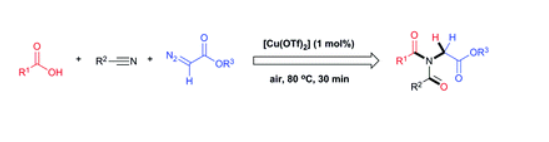
In situ generation of nitrilium from nitrile ylide and the subsequent Mumm rearrangement: copper-catalyzed synthesis of unsymmetrical diacylglycine esters
Jijun Chen,aYing Shao,abLiang Ma,aMeihua Maa and Xiaobing Wan*a(万小兵)
a Key Laboratory of Organic Synthesis of Jiangsu Province, College of Chemistry, Chemical Engineering and Materials Science, Soochow University, Suzhou 215123, China
bJiangsu Key Laboratory of Advanced Catalytic Materials and Technology, Advanced Catalysis and Green Manufacturing Collaborative Innovation Center, Changzhou University, Changzhou, China
Org. Biomol. Chem., 2016,14, 10723-10732
A novel in situ generation of nitrilium from a nitrile ylide and the subsequent Mumm rearrangement of carboxylic acid, nitrile, and diazo compounds gave various unsymmetrical diacylglycine esters in moderate to high yields. This copper-catalyzed cascade reaction enables one-pot generation of two C–N bonds, one C![]() O bond, and one C–H bond, with nitrogen as the only byproduct. The reaction has a broad functional-group tolerance, is rapid, easily scales up to the 100 mmol scale, and is insensitive to air and moisture.
O bond, and one C–H bond, with nitrogen as the only byproduct. The reaction has a broad functional-group tolerance, is rapid, easily scales up to the 100 mmol scale, and is insensitive to air and moisture.
链接:http://pubs.rsc.org/en/Content/ArticleLanding/2016/OB/C6OB02037B#!divAbstract
