aKey Laboratory of Organic Synthesis of Jiangsu Province, College of Chemistry, Chemical Engineering and Materials Science & Collaborative Innovation Center of Suzhou Nano Science and Technology, Soochow University, Suzhou
Org. Chem. Front., 2016,3, 1299-1303
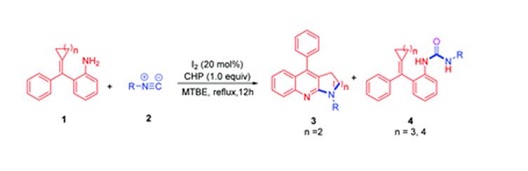
A new strategy for the catalytic synthesis of the pyrrolo[2,3-b]quinoline skeleton by the reaction of isocyanides with (cyclopropylidene(aryl)methyl)aniline (amino-MCPs) is presented, taking advantage of I2/CHP mediated carbodiimide intermediate generation. This reaction involves three C–N bonds and one C–C bond generation under metal-free and azide-free conditions.
链接:http://pubs.rsc.org/en/content/articlelanding/2016/qo/c6qo00373g#!divAbstract
