Mechanistic Insights into Photoredox/Nickel Dual-Catalyzed Difunctionalization of Alkynes: Revealing Roles of Noninnocent P-Ligand and Origins of Selectivities
Mengyu Xu1, Mengting Cao1, Rui Fu1, Xiaoguang Bao(鲍晓光)1,2*
1Innovation Center for Chemical Sciences, College of Chemistry, Chemical Engineering and Materials Science, Soochow University, 199 Ren-Ai Road, Suzhou Industrial Park, Suzhou, Jiangsu 215123, China
2Jiangsu Key Laboratory of Advanced Negative Carbon Technologies, Soochow University, Suzhou, Jiangsu 215123, China
J. Am. Chem. Soc. 2026, 148, 9608–9619
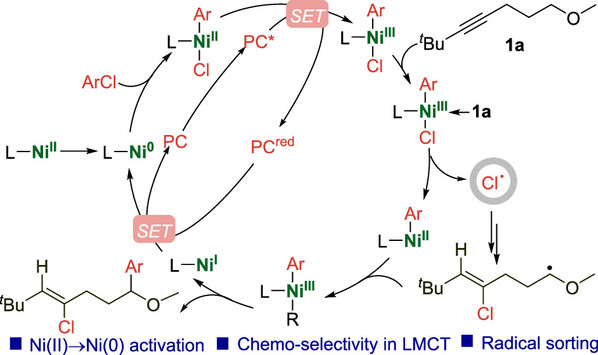
Abstract: Photoredox/nickel cooperative catalysis has advanced greatly to accomplish many challenging cross-coupling reactions. Nevertheless, the detailed mechanistic pathways of photoredox/nickel dual catalysis could be complicated in terms of conversions of oxidation states of the Ni complex and origins of chemo- and regioselectivities of products, which deserve further clarification. Herein, we conduct a combined computational and experimental study to gain mechanistic insights into the photoredox/Ni dual-catalyzed difunctionalization of alkynes. First, a novel route to transform the NiCl2(PCy3)2 precatalyst to a Ni(0) species is proposed under photoredox conditions. The noninnocent PCy3 ligand could play an important role in quenching the excited Ir(III) photocatalyst via a single-electron transfer (SET) step to yield a PCy3•+ species + Ir(II). Then, the reduced Ir(II) species could undergo a second SET with Ni(II) to afford a Ni(I) species. Meanwhile, the formed PCy3•+ species could combine with the hydroxide anion to yield a Cy3P(OH)• radical, which subsequently undergoes an H atom transfer step with the formed Ni(I) to furnish a Ni(0) species. In addition, the single-electron oxidation of Ni(II)ArCl by the excited photocatalyst to form a Ni(III)ArCl+ intermediate and the coordination of the alkynyl moiety of the substrate with the Ni(III) species are important factors to account for the chemoselective generation of Cl•, instead of Ar•, in the photoinduced ligand-to-metal charge transfer (LMCT) process. The generated Cl• could then undergo radical attack to the alkynyl moiety of the substrate, and a radical sorting mechanism is proposed to rationalize the origin of regioselectivity of the product.
Article information: https://doi.org/10.1021/jacs.5c20533
