Photoinduced Dearomative Oxyimination of Indoles Using Oxime Esters as Bifunctional Radical Precursors
YangMing1,2,ZiYou3(訾由)*, JiShun-Jun1,2(纪顺俊)*,XuXiao-Ping1,2,4(徐小平)*
1Key Laboratory of Organic Synthesis of Jiangsu Province, College of Chemistry, Chemical Engineering and Materials Science, Soochow University,Suzhou 215123, P. R. China
2Collaborative Innovation Center ofSuzhou Nano Science and Technology, Soochow University,Suzhou 215123, P. R. China
3School of Chemistry and Chemical Engineering,Nantong University, Nantong 226019, P. R. China
4Innovation Center for Chemical Science, Soochow University, Suzhou 215123, P. R.China
Org. Lett.2026, 28, 11, 3612–3618
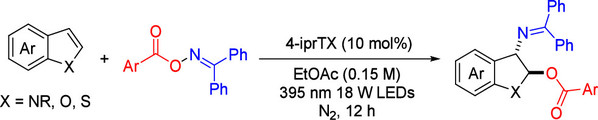
Abstract: A photoinduced, metal-free, and regioselective oxyimination of indole derivatives has been developed, enabling efficient difunctionalization via simultaneous C–O and C–N bond formation. Using oxime esters as dual radical precursors, this mild and sustainable strategy achieves a high atom economy and excellent diastereoselectivity. It can be applied to benzofuran and benzothiophene derivatives. This method provides a versatile and practical approach for dearomative transformations, offering new opportunities for the diastereoselective functionalization of indole derivatives.
Article information: https://doi.org/10.1021/acs.orglett.6c00690
