Organo-Catalyzed Asymmetric Michael–Hemiketalization–Oxa-Pictet–Spengler Cyclization for Bridged and Spiro Heterocyclic Skeletons: Oxocarbenium Ion as a Key Intermediate
Wei-Tai Fan†Nai-Kai Li†Lumei Xu‡Chunhua Qiao*‡(乔春华)Xing-Wang Wang*†(王兴旺)
†Key Laboratory of Organic Synthesis of Jiangsu Province, College of Chemistry, Chemical Engineering and Materials Science, and‡College of Pharmaceutical Science,Soochow University, Suzhou 215123,People’s Republic of China
Org. Lett.,2017,19(24), 6626--6629
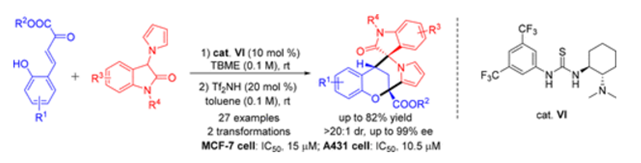
A Michael–hemiketalization–oxa-Pictet–Spengler cyclization has been developed for the construction of chiral bridged and spiro heterocyclic skeletons with one spiro stereogenic carbon center and two bridgehead carbon centers, utilizing cooperative catalysts of a Takemoto thiourea catalyst and a triflimide. In particular, an oxocarbenium ion acts as a key intermediate for this cyclization reaction. Additionally, biological evaluation of this type of novel structure has revealed obvious antiproliferative activity against some cancer cell lines.
链接:https://pubs.acs.org/doi/abs/10.1021/acs.orglett.7b03341
