Hyaluronic Acid-Shelled Disulfide-Cross-Linked Nanopolymersomes for Ultrahigh-Efficiency Reactive Encapsulation and CD44-Targeted Delivery of Mertansine Toxin
Yue Zhang†Kaiqi Wu†Huanli Sun*†(孙欢利)Jian Zhang†Jiandong Yuan‡Zhiyuan Zhong*†(钟志远)
†Biomedical Polymers Laboratory, and Jiangsu Key Laboratory of Advanced Functional Polymer Design and Application, College of Chemistry, Chemical Engineering and Materials Science,Soochow University, Suzhou 215123,P. R. China
‡BrightGene Bio-Medical Technology Co., Ltd., Suzhou 215123,P. R. China
ACS Appl. Mater. Interfaces,2018,10(2), 1597--1604
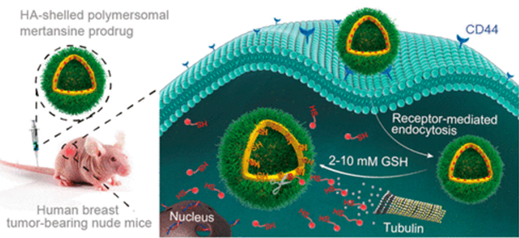
It was and remains a big challenge for cancer nanomedicines to achieve high and stable drug loading with fast drug release in the target cells. Here, we report on novel hyaluronic acid-shelled disulfide-cross-linked biodegradable polymersomes (HA-XPS) self-assembled from hyaluronic acid-b-poly(trimethylene carbonate-co-dithiolane trimethylene carbonate) diblock copolymer for ultrahigh-efficiency reactive encapsulation and CD44-targeted delivery of mertansine (DM1) toxin, a highly potent warhead for clinically used antibody-drug conjugates. Remarkably, HA-XPS showed quantitative encapsulation of DM1 even with a high drug loading content of 16.7 wt %. DM1-loaded HA-XPS (HA-XPS-DM1) presented a small size of ∼80 nm, low drug leakage under physiological conditions, and fast glutathione-triggered drug release. MTT assays revealed that HA-XPS was noncytotoxic while HA-XPS-DM1 was highly potent to MDA-MB-231 cells with an IC50comparable to that of free DM1. The in vitro and in vivo inhibition experiments indicated that HA-XPS could actively target MDA-MB-231 cells. Notably, HA-XPS-DM1 while causing little adverse effect could effectively inhibit tumor growth and significantly prolong survival time in MDA-MB-231 human breast tumor-bearing mice. HA-XPS-DM1 provides a novel and unique treatment for CD44-positive cancers.
链接:https://pubs.acs.org/doi/10.1021/acsami.7b17718
