Ligand-Controlled Copper(I)-Catalyzed Cross-Coupling of Secondary and Primary Alcohols to α-Alkylated Ketones, Pyridines, and Quinolines
Da-Wei Tan†Hong-Xi Li*†‡(李红喜)Da-Liang Zhu†Hai-Yan Li†David James Young§Jian-Lin Yao*†(姚建林)Jian-Ping Lang*†‡(郎建平)
†College of Chemistry, Chemical Engineering and Materials Science,Soochow University, Suzhou 215123,China
‡State Key Laboratory of Organometallic Chemistry, Shanghai Institute of Organic Chemistry,Chinese Academy of Sciences, Shanghai 200032,China
§Faculty of Science, Health, Education and Engineering,University of the Sunshine Coast, Maroochydore DC, Queensland 4558,Australia
Org. Lett.,2018,20(3), 608--611
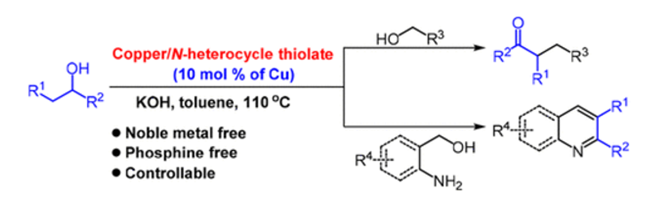
One hexanuclear Cu(I) cluster of 4,6-dimethylpyrimidine-2-thiolate efficiently catalyzes the dehydrogenative cross-coupling of secondary and primary alcohols to α-alkylated ketones with high selectivity. This transformation proceeds through a one-pot sequence of dehydrogenation of alcohols, condensation of aldehydes and ketones, hydrogenation of the resulting α,β-unsaturated ketones, and dehydrogenation of the α-alkylated alcohols to generate α-alkylated ketones. This catalytic system also displays high activity for the annulation reaction of secondary alcohols with γ-amino- and 2-aminobenzyl alcohols to yield pyridines and quinolines, respectively.
链接:https://pubs.acs.org/doi/10.1021/acs.orglett.7b03726
