Capture of SO3 isomers in the oxidation of sulfur monoxide with molecular oxygen
Zhuang Wu a, Bo Lu a, Ruijuan Feng a, Jian Xu a, Yan Lu a, Huabin Wan a, Andre´ K. Eckhardt b, Peter R. Schreiner b, Changjian Xie c, Hua Guo c and Xiaoqing Zeng *(曾小庆)
a College of Chemistry, Chemical Engineering and Materials Science,
Soochow University, Suzhou 215123, China.
b Institute of Organic Chemistry, Justus-Liebig University, Heinrich-Buff-Ring 17,35392 Giessen, Germany
c Department of Chemistry and Chemical Biology, University of New Mexico,Albuquerque, NM 87131, USA
Chem. Commun., 2018, 54, 1690--1693
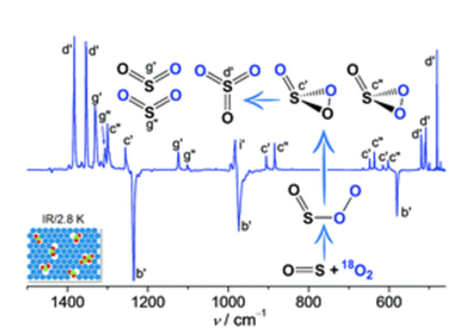
When mixing SO with O2in N2, Ne, or Ar, an end-on complex OS–OO forms in the gas phase and can subsequently be trapped at cryogenic temperatures (2.8–15.0 K). Upon infrared light irradiation, OS–OO converts to SO3and SO2+ O with the concomitant formation of a rare 1,2,3-dioxathiirane 2-oxide,i.e., cyclic OS(
链接:http://pubs.rsc.org/en/Content/ArticleLanding/2018/CC/C7CC09389F#!divAbstract
