Shuwei Lin, Yi Li, Baozong Li, and Yonggang Yang*(杨永刚)
Jiangsu Key Laboratory of Advanced Functional Polymer Design and Application, Department of Polymer Science and Engineering, State and Local Joint Engineering Laboratory for Novel Functional Polymeric Materials, College of Chemistry, Chemical Engineering, and Materials Science, Soochow University, Suzhou 215123, People’s Republic of China
Langmuir, 2016, 32 (29), pp 7420–7426
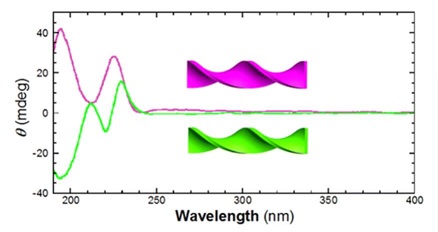
Eight dipeptides, composed of phenylalanine and phenylglycine, that are able to self-assemble into twisted nanoribbons in deionized water are synthesized. The handedness of the nanoribbons is controlled by the chirality of the phenylalanine and the steric hindrance owing to the phenyl group of the phenylglycine. When the phenylalanine is at the C-terminal, π–π stacking by the phenyl groups, hydrogen bonding by the NH group of the phenylalanine, and hydrophobic associations of the alkyl chains control the stacking of the molecules. When phenylglycine is at the C-terminal, the chiral π–π stacking by the phenyl groups of the phenylalanines is suppressed. The hydrogen bonds formed by the NH groups of the phenylalanines had a greater contribution on forming organic self-assemblies than those formed by the NH groups of the phenylglycines.
链接:http://pubs.acs.org/doi/abs/10.1021/acs.langmuir.6b01874
