Degradation of ibuprofen in water by FeII-NTA complex-activated persulfate with hydroxylamine at neutral pH
Yaoyao Jin, Sheng-Peng Sun, Xueying Yang, Xiao Dong Chen
Suzhou Key Laboratory of Green Chemical Engineering, School of Chemical and Environmental Engineering, College of Chemistry, Chemical Engineering and Materials
Science, Soochow University, Suzhou, Jiangsu 215123, PR China
Chemical Engineering Journal 337 (2018) 152--160
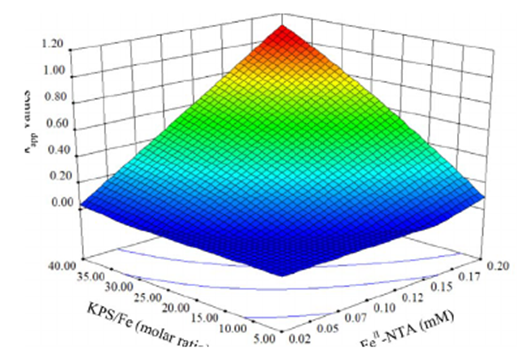
The activation of potassium persulfate (KPS) by FeII-NTA complex has been investigated to degrade ibuprofen in aqueous solutions under neutral pH conditions. The results showed that ibuprofen degradation by FeII-NTA/KPS process was not successful due to the rapid oxidation of FeII-NTA complex by dissolved oxygen. However, an efficient degradation of ibuprofen was achieved by FeII-NTA/KPS process in the presence of hydroxylamine (HA). It was evident that the steady-state FeIIconcentration in FeII-NTA/HA/KPS process was much higher than that in FeII-NTA/KPS process because of the quick regeneration of FeII-NTA complex by HA. The suitable conditions for FeII-NTA/HA/KPS process were obtained at [FeII] = 0.1 mM, [NTA/Fe] = 1:1, [HA/Fe] = 20:1 and [KPS/Fe] = 30:1. The generation of sulfate radical (SO4
链接:https://www.sciencedirect.com/science/article/pii/S1385894717322167?via%3Dihub
