Highly Site-Selective Formation of Perfluoroalkylated Anilids via a Protecting Strategy by Molybdenum Hexacarbonyl Catalyst
Chunchen Yuan, Ping Dai, Xiaoguang Bao*(鲍晓光), and Yingsheng Zhao*(赵应声)
Key Laboratory of Organic Synthesis of Jiangsu Province, College of Chemistry, Chemical Engineering and Materials Science, Soochow University, Suzhou 215123, China
Org. Lett.2019,21, 6481--6484
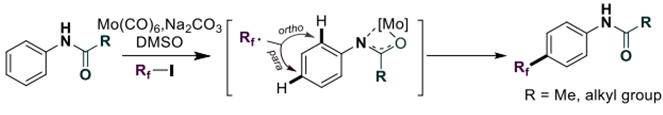
Introducing a perfluoroalkyl group on the aromatic ring with high site selectivity remains a challenging area in organofluorine chemistry. We herein report a highly para-selective C–H perfluoroalkylation of aniline substrates using the molybdenum hexacarbonyl catalyst. Various substituted anilids derived from anilids were well-tolerated, affording the corresponding products in moderate to good yields. Preliminary mechanism studies and density functional theory calculations revealed the coordination of Mo catalyst with amides as the key factor to realize para selectivity.
链接:https://pubs.acs.org/doi/10.1021/acs.orglett.9b02362
