MicroRNA-Catalyzed Cancer Therapeutics Based on DNA-Programmed Nanoparticle Complex
†††‡§††††*†(马楠)
† The Key Lab of Health Chemistry and Molecular Diagnosis of Suzhou, College of Chemistry, Chemical Engineering and Materials Science, Soochow University, Suzhou, 215123, P. R. China
‡ Department of Chemistry, Hong Kong Branch of Chinese National Engineering Research Center for Tissue Restoration and Reconstruction, Institute of Molecular Functional Materials, Institute for Advanced Study, State Key Laboratory of Neuroscience, Division of Biomedical Engineering, and Division of Life Science, The Hong Kong University of Science and Technology, Clear Water Bay, Kowloon, Hong Kong P. R. China
§HKUST-Shenzhen Research Institute, Shenzhen 518057, P. R. China
ACS Appl. Mater. Interfaces, 2017, 9 (39), 33624--33631
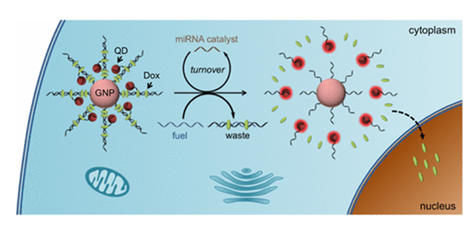
The use of cancer-relevant microRNA molecules as endogenous drug release stimuli is promising for personalized cancer treatment yet remains a great challenge because of their low abundance. Herein, we report a new type of microRNA-catalyzed drug release system based on DNA-programmed gold nanoparticle (GNP)–quantum dot (QD) complex. We show that a trace amount of miRNA-21 molecules could specifically catalyze the disassembly of doxorubicin (Dox)-loaded GNP–QDs complex through entropy driven process, during which the Dox-intercalating sites are destructed for drug release. This catalytic reaction could proceed both in fixed cells and live cells with miRNA-21 overexpression. Dox molecules could be efficiently released in the cells and translocate to cell nuclei. QD photoluminescence is simultaneously activated during catalytic disassembly process, thus providing a reliable feedback for microRNA-triggered drug release. The GNP–QDs–Dox complex exhibits much higher drug potency than free Dox molecules, and therefore represents a promising platform for accurate and effective cancer cell treatment.
链接:http://pubs.acs.org/doi/10.1021/acsami.7b09420
